Contact Information: Media Contact: Amy Cook 925.552.7893 Email Contact
Carl Zeiss Meditec Enhances Clinical Value by Offering Solutions for Cataract and Refractive Surgery
Company to Showcase New, Innovative Technologies at ASCRS
| Source: Carl Zeiss Meditec, Inc.
SAN DIEGO, CA -- (MARKET WIRE) -- April 30, 2007 -- ASCRS Booth # 2641 Hall D -- Carl Zeiss
Meditec, Inc. will demonstrate its newest technologies and gold-standard
products here at the American Society of Cataract and Refractive Surgery
(ASCRS) meeting in San Diego, April 28 - May 1, 2007. The company's
platforms and technological advancements address a broad spectrum of
applications designed to improve workflow and enhance patient management.
"Our newest initiatives coupled with recent advances to existing product
platforms reflect our focused commitment to the continuous development and
enhancement of technical capabilities," said Jim Taylor, president and
chief executive officer of Carl Zeiss Meditec, Inc. "Our platforms are
designed not only to offer solutions for today's urgent clinical demands,
but also with a vision for future needs."
Product Platforms to be Highlighted
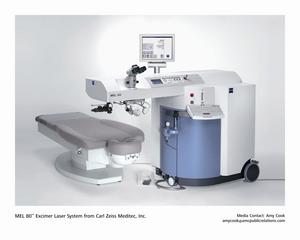
Offering refractive laser systems for the effective and patient-friendly
correction of vision defects, the company will demonstrate its MEL 80™
excimer system and the VISUMAX™ femtosecond laser system, which recently
received U.S. Food & Drug Administration (FDA) clearance. Clinical trials
for the MEL 80 showed that 93 percent of patients were corrected at three
months to 20/20 or better visual acuity, and 41 percent were corrected to
20/12.5 or better at six months. Additionally, 85 percent of the patients
achieved within one-half of a diopter of the intended correction at the
three month post-operative follow-up. The combination of both platform
lasers, including the pivoting patient bed and integrated data management
system, will enable physicians to complete a full refractive procedure in
one location without the need to move the patient, or perform redundant
data entries.
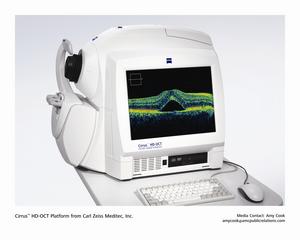
Joining the market-leading Stratus OCT, the company will demonstrate its
latest advancement in spectral domain OCT with the Cirrus™ HD-OCT
platform. This high-end instrument is ideal for the most demanding retinal
cases because it offers proprietary high definition technology for superior
imaging and visualization of the retinal tissue. This high definition
spectral domain technology has faster scanning capabilities, capturing more
data and revealing greater detail of retinal tissue and pathology.
Additionally, the Cirrus system's high precision measurements facilitate
structural evaluation and monitoring.
The company will also showcase the newest applications for the Stratus
OCT™ platform, which is recognized as the standard-of-care instrument
supporting comprehensive eye-care professionals and retinal specialists
worldwide. The in-vivo cross sectional images and quantitative analysis of
the retina help to facilitate a more complete assessment of cataract
patients pre- and post-operatively, as well as patients with glaucoma and
retinal disease for enhanced patient management.
Further evidence of CZM's commitment to OCT technology includes the Visante
OCT -- the first stand-alone, high resolution OCT system for the anterior
segment of the eye, including the cornea, iris, angle and the lens. The
company will introduce new refractive tools for surgical planning along
with its latest iridocorneal tools for glaucoma patient assessment.
Carl Zeiss Meditec has been leading the development of progression analysis
for more than 20 years, offering significant clinical benefits that
ultimately increase the potential for correct diagnoses. The new Guided
Progression Analysis™ (GPA) tools for the GDx Scanning Laser Polarimeter
and Stratus OCT, coupled with the enhanced tools for the Humphrey® Field
Analyzer, will help doctors to make accurate, individual treatment
decisions.
The company's suite of platforms also supports today's connectivity needs
with a pathway for the electronic workflow demands of the future. The new
VISUPAC Star Storage and Review software is the single platform that
organizes all Carl Zeiss Meditec images and print data into a
patient-centric database. With improved access to current and past data,
patient consultation is enhanced and workflow is considerably improved on a
daily basis. In addition, the VISUPAC Star software supports EMR
integration; therefore standardized interface protocols are available to
all EMR vendors.
To see a demonstration of these products and learn more about the company,
please stop by the Carl Zeiss Meditec booth at ASCRS (Booth #2641 Hall D)
or visit www.meditec.zeiss.com/usa.
About Carl Zeiss Meditec
Carl Zeiss Meditec AG (ISIN: DE 0005313704) is one of the world's leading
medical technology companies. This market position is based on over 160
years of experience in optical innovation. The company has two primary
areas of activity. In the field of ophthalmology, Carl Zeiss Meditec offers
integrated solutions for treating the four main eye diseases: vision
defects (refraction), cataracts, glaucoma and retinal disorders. Product
innovations range from basic systems such as slit lamps and fundus cameras
to standard setting diagnostic systems such as the Humphrey® Field
Analyzer, the Stratus OCT™ and the IOLMaster®, through to the surgical
microscopes and innovative treatment systems in refractive laser surgery.
In the field of neuro and ENT surgery, Carl Zeiss Meditec is the world's
leading provider of surgical microscopes and microsurgical visualization
solutions for a very broad range of applications, such as tumour and
vascular surgery in the head region and/or spinal surgery. The most recent
example of our innovative performance in the area of microsurgery is the
OPMI Pentero® visualization system, which allows efficient and ergonomic
patient treatment.
Carl Zeiss Meditec's medical technology portfolio is rounded off by
visualization systems for doctors in private practice and promising future
technologies such as intraoperative radiation therapy, which allows the
targeted treatment of breast cancer and brain cancer directly during
surgery.
Carl Zeiss Meditec AG is based in Jena, Germany, with subsidiaries in
Germany (Carl Zeiss Meditec Surgical GmbH and Carl Zeiss Meditec
Vertriebsgesellschaf mbH), the USA (Carl Zeiss Meditec, Inc., Dublin,
California), in Japan (Carl Zeiss Meditec Co., Ltd., Tokyo), Spain (Carl
Zeiss Meditec Iberia S.A., Madrid) and France (Ioltech SAS, La Rochelle,
and Carl Zeiss Meditec France SAS, Le Pecq).
Thirty-five percent of the Carl Zeiss Meditec shares are in free float. The
remaining 65 percent are held by Carl Zeiss, one of the world's leading
international groups engaged in the optical and opto-electronics industry.
For more information, visit www.meditec.zeiss.com/usa.