ATLANTA, GA--(Marketwired - Jan 12, 2015) - GeoVax Labs, Inc. (
In October 2014, GeoVax announced its initiation of a new program for the development of vaccines to prevent Ebola virus infection. GeoVax is developing two Ebola vaccines, GOVX-E301 and GOVX-E302. GOVX-E301 is being developed as a monovalent vaccine for the Zaire strain of Ebola, the virus responsible for the current outbreak. GOVX-E302 is being developed as a trivalent vaccine for protection against all three versions of Ebola known to be lethal in humans (Zaire, Sudan and Bundibugyo). Both GOVX-E301 and GOVX-E302 utilize GeoVax's recombinant modified vaccinia Ankara (MVA) platform to expresses virus-like particles (VLPs) in the person being vaccinated.
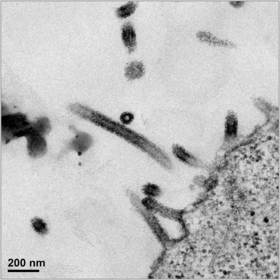
Since initiation of its Ebola vaccine program, GeoVax has successfully designed its MVA-based vaccine vector for the Zaire strain of Ebola using the most current genetic sequences from the ongoing epidemic. The Company has now demonstrated that its vaccine produces VLPs in vaccine-expressing human cells, an important milestone in the vaccine's development. GeoVax believes that MVA-expressed VLPs, which display the virus's surface protein (glycoprotein), will be highly effective at eliciting antibody capable of preventing and controlling Ebola virus infections. The electron-micrograph pictured shows cultured human cells expressing the Zaire Ebola VLPs.
GeoVax's goals for 2015 include completion of initial immunogenicity studies in small animals, followed by proof-of-concept challenge studies. The Company expects to begin challenge studies in non-human primates in late 2015. Simultaneously, GeoVax will be advancing its vaccine manufacturing and test methods, with the goal of initiating human clinical testing in 2016.
Robert McNally, PhD, President & CEO of GeoVax commented, "I am very pleased with the rate of our progress for our Ebola program. Demonstrating that our vaccine effectively produces virus-like particles is a significant milestone, and our scientific staff is working hard to advance the program through the requisite testing."
Dr. McNally added, "The Ebola epidemic continues to receive worldwide attention, and a number of companies are working to develop vaccines. However, as we've seen already with at least one company's efforts, vaccine safety issues have arisen, while other companies plan to use two different vaccines to hopefully achieve adequate immune responses. With the safety and efficacy track record we have established for our vaccine delivery platform through our HIV program, and with the use of the most current genomic sequences of the Ebola virus, GeoVax has a high probability of success in developing a vaccine superior to the vaccines currently entering clinical trials. Additionally, our focus on a trivalent Ebola vaccine against multiple strains of the virus is something the world needs, as history has shown that the current outbreak will not be the last."
Dr. McNally concluded, "Historically, GeoVax has been identified as an HIV vaccine development company. However, with the current Ebola epidemic being suited to our technology, we believe it is a natural expansion of GeoVax's product pipeline. As our HIV vaccine program continues to be supported by the National Institutes of Health (NIH), we have now made our Ebola program a top priority for allocation of internal resources. As this new program develops, we intend to explore other areas where our technology and expertise may be applicable. We look forward to updating our shareholders as we move ahead."
About GeoVax
GeoVax Labs, Inc. (
All of the clinical trials for GeoVax's preventive HIV vaccines have been conducted by the HIV Vaccine Trials Network with funding from the NIH. Overall, GeoVax's HIV vaccines, in various doses and combinations, have been tested in close to 500 humans. The Company's proprietary Ebola vaccine technology has been developed internally and its HIV vaccine technology was developed in collaboration with Emory University, the NIH, and the U.S. Centers for Disease Control and Prevention (CDC). For more information, go to www.geovax.com.
Forward-Looking Statements
Certain statements in this document are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act. These statements are based on management's current expectations and are subject to uncertainty and changes in circumstances. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax can develop and manufacture its vaccines with the desired characteristics in a timely manner, GeoVax's vaccines will be safe for human use, GeoVax's vaccines will effectively prevent HIV or Ebola infection in humans, vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete vaccine development, there is development of competitive products that may be more effective or easier to use than GeoVax's products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control. GeoVax assumes no obligation to update these forward-looking statements, and does not intend to do so. More information about these factors is contained in GeoVax's filings with the Securities and Exchange Commission including those set forth at "Risk Factors" in GeoVax's Form 10-K.
Contact Information:
Contact:
Adam S. Holdsworth
ProActive Capital
646-862-4607
www.ProActiveCapital.com