ATLANTA, GA--(Marketwire - May 3, 2011) - One of the greatest problems facing GI endoscopy labs today is the need to maintain proper procedures and protocols to ensure patient safety and maintain accreditation. EndoChoice will be showcasing two tools that assist with attaining those goals at the 38th Annual Course of the Society of Gastroenterology Nurses and Associates (SGNA) held from May 8-10th in Indianapolis, Indiana.
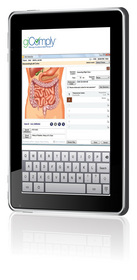
The company will debut giComply™, a software program that automates the traditional pathology logbook and generates fast, accurate requisitions and labels. With giComply, the nurse selects a patient from the practice management system; enters specimen information using intuitive, onscreen anatomic graphics and drop-down menus, then prints the requisition and labels. All information is automatically entered into the electronic logbook which includes a searchable database and the ability to color-code entries for faster, easier reference.
"In talking with our customers, we discovered that maintaining a logbook was one of their headaches, but a headache that could have substantial impact on the practice's accreditation," explained Mark Gilreath, EndoChoice Chief Executive Officer and founder. "They also described the difficulty in legibly completing requisitions and trying to complete labels on the side of specimen jars. giComply addresses these problems while streamlining workflow and ensuring accuracy."
EndoChoice will also be featuring its new Compliance EndoKits®, a solution that addresses the #1 health technology risk today, "reprocessing of flexible endoscopes," as cited by the ECRI Institute. Human variability is one of the biggest factors in breaches of infection control protocols when handling and reprocessing flexible endoscopes. Accreditation groups issue standards and guidelines for mitigating these risks, but compliance with the measures often varies based on staff and budget constraints. EndoChoice helps drive widespread compliance with these standards by providing GI professionals with the critical supplies needed to help reduce cross-contamination risks at affordable prices.
About EndoChoice
EndoChoice is a global leader and medical technology provider for specialists treating a wide range of gastrointestinal diseases. EndoChoice is committed to the GI endoscopy market offering a comprehensive portfolio of treatment options for tissue acquisition, anatomic pathology, hemostasis, luminal patency, retrieval, infection control and procedural support. For more information visit